The Ministry of Public Health deceived the Legislative Power by responding to a request for reports with data extracted from the website of the tobacco company Philip Morris, instead of sending the answer that the technical services of the Ministry had given.
On April 15, 2021, deputy Lucía Etcheverry (MPP-FA) sent the MPH a request for reports on the approval of decree 87/021, which enabled the marketing of heated tobacco products (HTP), which had been expressly prohibited since 2009. The presidential decision breached the Framework Convention on Tobacco Control (FCTC) of the World Health Organization (WHO), which the country ratified through Law No. 17,793.
In her request, the legislator asked five questions:
1- What scientific evidence, reports or scientific investigations were taken as the basis for the issuance of Decree of the Executive Power No. 87/021, of March 3, 2021, related to Decree No. 534/009, of November 23, 2009.
2- What evidence and scientific report did you have as support to affirm in the Results of Decree No. 87/021 that ‘there are electronic devices for the administration of nicotine that use a technology through which dry tobacco is heated, for which there are scientific data indicating that they result in less exposure of users to toxic substances associated with traditional tobacco consumption’.
3- If, prior to the issuance of Decree No. 87/021, the Department of Pulmonology, Cardiology and Oncology of the Faculty of Medicine of the University of the Republic was consulted.
4- If, prior to the issuance of the aforementioned decree, the Pan American Health Organization was consulted.
5 – If, prior to the issuance of the aforementioned Decree, the authorities of that Ministry met with representatives of the companies that produce and market tobacco products and with the companies that market and import electronic devices for smoking.
Upon receiving the query, the General Directorate of Health of the MPH gave the procedure the number 3/2754/2021 and decided to consult the technical services of the Ministry, in this case the National Program Against Tobacco (NPAT), which is directed by the doctor Elba Esteves. The internal request for information was sent on Friday, April 27, and Esteves’ response arrived three days later, on Monday, April 30.
“The NPAT did not participate in the proposal, analysis, or preparation of Decree 87/021, it became aware of it on March 19, 2021 due to an unofficial communication,” explained the expert, according to the documentation accessed by Sudestada.
Regarding Etcheverry’s first question, Esteves replied that «The PNCT is not aware of said evidence or report.»
In response to the second query, he replied that «The PNCT does not have information to support the aforementioned statement» that the devices imply less exposure for users.
As for the eventual requests for advice to the medical chairs and to PAHO, the only response was: «It is unknown if these consultations were carried out.»
Finally, the MPH specialist explained that «The PNCT did not hold any meeting or contact of any kind with representatives of the aforementioned companies» in relation to the approval of Decree No. 87/021.
In addition, he explained that, as mandated by article 5.3 of the WHO Framework Convention on Tobacco Control, of which Uruguay is a signatory, the FCTC «has a procedural protocol for meetings with tobacco companies, which establishes the need for a request for a meeting with an explanation of the reason for the meeting. If the reason is exclusively related to the application of Law 18,256 and its modifications, the meeting is held with the presence of a representative of civil society and a meeting Minutes is drawn up.
The response of the MPH expert on tobacco was not to the liking of the ministerial authorities, and it was archived. Instead, file No. 12/001/3/4526/2021 was opened and the Legal Services Division of the Ministry prepared another version more adjusted to the interests of the Executive Branch and closer to the tobacco industry.
Apparently, the text took a long time to be drafted, and only reached parliament on July 22.
A response copied from the Internet
The response sent does not refer to any of the five questions raised by the representative Etcheverry, and recognizes that it is a «partial» response. The first clarification made is that the decree was approved by the Council of Ministers and that it was «substantiated in the sphere of the Presidency of the Republic.»
Immediately, the text sent to parliament begins to talk about the Food and Drug Administration (FDA) of the United States, a country that did not sign the WHO FCTC, and about whose decisions the legislator had not asked.
Thus, the MPH first refers to an article on the FDA website in which the difference between traditional and emerging cigarettes is explained, but it omits to reproduce some sections of the resolution that are critical of the product.
“There is no safe tobacco product. Non-combustible cigarettes can help reduce the risks of harm associated with tobacco for adult smokers who switch completely from combustion cigarettes”, says the Uruguayan ministry in its citation. However, it hides the rest of the sentence: «but all tobacco products can lead to nicotine addiction and contain toxic chemicals that cause cancer and can cause serious health problems.»
As a complement to its statement, the MPH places a link to the FDA website, but does not provide a translation of the article, which is striking in the case of an official response from one branch to another of the Uruguayan State.
A second reference to the FDA – with the corresponding link – is made in the following paragraph. It refers to the authorization of the sale of the device called IQOS, which is manufactured by the company Philip Morris International. At no time does the MPH refer to the devices of other companies such as British American Tobacco (BAT), Japan Tobacco International or Pax Labs, which also market these products.
The explanation for this exclusive mention can be found in the fact that from this fourth page of the document, all the references are taken from inputs made by the Philip Morris tobacco company.
Indeed, one of the sources of inspiration is a web page -which is in English- titled «Growing scientific and regulatory consensus on tobacco harm reduction» and reports that, according to PMI, «several governments recognize that smoke-free products have a role to play in reducing the harm caused by smoking and have adopted a tobacco harm reduction approach.”
The link copied by the MPH and included in the response to representativeEtcheverry is found in the following section of the PMI website:

The third reference to the FDA decisions has to do with the “modified risk” declaration of these devices, one of the plot tricks of the industry to say that its products are less harmful than traditional ones.
This decision of the United States government agency also appears on the Philip Morris website, in the following section:

Then, and without referring to any of the questions included in the parliamentary request, the MPH adds that “there are numerous international scientific publications available to the general public that describe reduced exposure to harmful substances and potentially harmful” in the products authorized by the decree of March 2021.
Then, it provides four examples of those references, and again includes addresses to web pages that are in English, without any official translation. It is logical, because several of these references were also taken from the Philip Morris website, according to Sudestada.
The first is a decision by the Netherlands National Institute for Public Health and the Environment entitled “A method for comparing the carcinogenicity impact of tobacco products: a case study of heated tobacco versus cigarettes. Risk analysis». In this case, the address included in the MPH (https://doi.org/10.1111/risa.13482) is a documentary identification obtained from the online bookstore used by the company.
This reference is found in the next section of the PMI page.

The second academic reference cited by the MPH in its response to the Legislative Branch deals with “Carbon monoxide levels after inhalation of heated tobacco products’ new generation”, but this work does not appear on the company’s website.
However, it does appear as a footnote on page 12 of the March 2019 issue of the journal PMI Science, a publication that the tobacco company publishes with the following purpose: “to provide an overview of the most recent scientific developments behind PMI’s approach to achieving a smoke-free future through a range of alternatives to cigarettes that do not burn tobacco.»
The third scientific work cited is on the PMI website and belongs to the German Federal Institute for Risk Analysis. The study deals with “Levels of selected analytes in emissions of Heated Tobacco products relevant to the determination of risks to humans”. In this case, the academic articles are the same, but the hyperlinks are different.

The fifth link that appears on the Philip Morris company website refers to the Committee on Toxicity of Chemicals in Food, Consumer Products and the UK Environment, but it is a pronouncement and not a scientific study. The document is entitled «Declaration on the toxicological evaluation of new heating products without tobacco combustion», from the year 2017. In the latter case, the link coincided again.
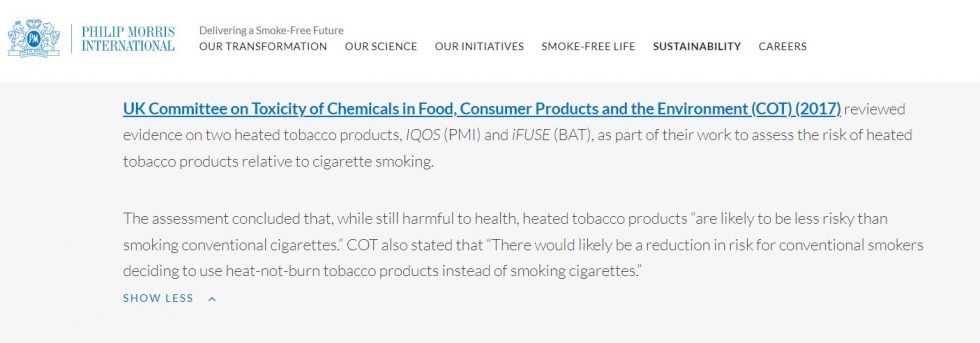
The response to the request for information that, indirectly, helped prepare the Philip Morris tobacco company, ends with a curious statement: “Having given partial response to what was requested, based on the information available in this Ministry, is submitted suggesting the referral of these actions to the National Parliament”.
Sudestada tried to consult the director of the National Program Against Tobacco, Elba Esteves, the general director of Health Miguel Asqueta and the minister Daniel Salinas, but none of the three interviews was possible.




